Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Sr
Sr MnO
MnO
Kajimoto, Ryoichi; Yoshizawa, Hideki*; Kawasaki, Ryuichi*; Noda, Kohei*; Kuwahara, Hideki*
Journal of the Physical Society of Japan, 74(1), p.502 - 503, 2005/01
Times Cited Count:2 Percentile:19.73(Physics, Multidisciplinary)The spin wave dispersion relation of the C-type antiferromagnetic structure in Nd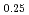 Sr
Sr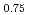 MnO
MnO was determined by neutron scattering. The spin exchange interactions show a large anisotropy, which evidences the d
was determined by neutron scattering. The spin exchange interactions show a large anisotropy, which evidences the d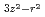 orbital ordering along the
orbital ordering along the 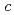 axis. The exchange interaction along the
axis. The exchange interaction along the 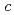 direction is fairly large and similar to the value in the ferromagnetic metallic phase, suggesting there is finite hopping of charges along the orbital chains.
direction is fairly large and similar to the value in the ferromagnetic metallic phase, suggesting there is finite hopping of charges along the orbital chains.
 studied by resonant inelastic X-ray scattering
studied by resonant inelastic X-ray scatteringInami, Toshiya; Fukuda, Tatsuo; Mizuki, Junichiro; Ishihara, Sumio*; Kondo, Hiroshi*; Nakao, Hironori*; Matsumura, Takeshi*; Hirota, Kazuma*; Murakami, Yoichi*; Maekawa, Sadamichi*; et al.
Physical Review B, 67(4), p.045108_1 - 045108_6, 2003/01
Times Cited Count:56 Percentile:87.92(Materials Science, Multidisciplinary)We report resonant inelastic x-ray scattering experiments of the orbitally ordered manganite LaMnO . When incident photon energy is tuned near the Mn
. When incident photon energy is tuned near the Mn  absorption edge, the spectra reveal three features at 2.5 eV, 8 eV and 11 eV. The 8 eV and 11 eV peaks are ascribed to transitions from the O 2
absorption edge, the spectra reveal three features at 2.5 eV, 8 eV and 11 eV. The 8 eV and 11 eV peaks are ascribed to transitions from the O 2 bands to the empty Mn 3
bands to the empty Mn 3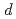 and
and 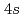 /
/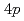 bands, respectively. On the other hand, the 2.5 eV peak is considered an orbital excitation across the Mott gap,
bands, respectively. On the other hand, the 2.5 eV peak is considered an orbital excitation across the Mott gap, 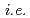 an electron excitation from the effective lower Hubbard band with the
an electron excitation from the effective lower Hubbard band with the 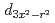 and
and 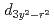 orbital characters to the upper Hubbard band with the
orbital characters to the upper Hubbard band with the 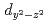 and
and 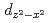 ones. The weak dispersion and characteristic azimuthal angle dependence of this excitation are well reproduced by a theory which includes orbital degeneracy and strong electron correlation.
ones. The weak dispersion and characteristic azimuthal angle dependence of this excitation are well reproduced by a theory which includes orbital degeneracy and strong electron correlation.
 solutions
solutionsHaba, Hiromitsu; Tsukada, Kazuaki; Asai, Masato; Goto, Shinichi*; Toyoshima, Atsushi; Nishinaka, Ichiro; Akiyama, Kazuhiko; Hirata, Masaru; Ichikawa, Shinichi; Nagame, Yuichiro; et al.
Journal of Nuclear and Radiochemical Sciences, 3(1), p.143 - 146, 2002/06
We have investigated the sorption behavior of element 104 rutherfordium (Rf) on an anion exchange resin from HCl and HNO solutions. In the HCl experiments, the distribution coefficients of Rf increase with an increase of HCl concentration from 7.0 M to 11.5 M, indicating that anionic species such as [Rf(OH)Cl
solutions. In the HCl experiments, the distribution coefficients of Rf increase with an increase of HCl concentration from 7.0 M to 11.5 M, indicating that anionic species such as [Rf(OH)Cl ]
] or [RfCl
or [RfCl ]
] are formed. This sorption behavior of Rf is typical of the group-4 elements Zr and Hf, and is quite different from that of the pseudo-homologue Th. It is also noted that the distribution coefficients decrease in the order Rf, Zr, Hf at 9.0 M HCl, which is consistent with the expected order of ionic radii. On the other hand, Rf appears to behave like Zr and Hf in 8 M HNO
are formed. This sorption behavior of Rf is typical of the group-4 elements Zr and Hf, and is quite different from that of the pseudo-homologue Th. It is also noted that the distribution coefficients decrease in the order Rf, Zr, Hf at 9.0 M HCl, which is consistent with the expected order of ionic radii. On the other hand, Rf appears to behave like Zr and Hf in 8 M HNO not like Pu and Th.
not like Pu and Th.
 Ca
Ca MnO
MnO system as 'Magnetic Relaxor'
system as 'Magnetic Relaxor'Yamada, Yasusada; Iwase, Takeshi*; Takakura, Tomoyuki*; Suzuki, Junichi; Wakahiki, Masaya*
Journal of the Physical Society of Japan, Vol.70, Supplement A, p.70 - 72, 2001/00
no abstracts in English
 Sr
Sr MnO
MnO (x
(x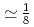 )
)Yamada, Yasusada; Suzuki, Junichi; Oikawa, Kenichi*; Katano, Susumu; Fernandez-Baca, J. A.*
Physical Review B, 62(17), p.11600 - 11608, 2000/11
Times Cited Count:38 Percentile:82.95(Materials Science, Multidisciplinary)no abstracts in English
 Sr
Sr MnO
MnO (x
(x 0.12)
0.12)Inami, Toshiya; Ikeda, Naoshi*; Murakami, Yoichi*; Koyama, Ichiro*; Wakabayashi, Yusuke*; Yamada, Yasusada*
Japanese Journal of Applied Physics, 38(suppl.38-1), p.212 - 214, 1999/06
Times Cited Count:15 Percentile:65.98(Physics, Applied)no abstracts in English
Journal of Nuclear Materials, 254, p.14 - 21, 1998/00
Times Cited Count:16 Percentile:76.3(Materials Science, Multidisciplinary)no abstracts in English
 B
B MnO
MnO (A:La,Pr;B:Sr,Ca)
(A:La,Pr;B:Sr,Ca)Inami, Toshiya*; Hino, O.*; Nodo, S.*; Kaneo, Rinna*; Ikeda, Naoshi*; Yamada, Yasusada*; Katano, Susumu
Physica B; Condensed Matter, 241-243, p.433 - 435, 1998/00
Times Cited Count:6 Percentile:42.67(Physics, Condensed Matter)no abstracts in English
 Sr
Sr MnO
MnO
Yamada, Yasusada*; Hino, O.*; Nodo, S.*; Kaneo, Rinna*; Inami, Toshiya*; Katano, Susumu
Physical Review Letters, 77(5), p.904 - 907, 1996/07
Times Cited Count:315 Percentile:98.66(Physics, Multidisciplinary)no abstracts in English
Kume, Tamikazu
Riken Shimpojiumu Kasokuki O Mochiita Yodenshi Riyo Gijutsu No Tenkai, 0, p.19 - 22, 1996/00
no abstracts in English
Abe, Hitoshi; Usuda, Shigekazu; Takeishi, Hideyo; Tachimori, Shoichi
J. Liquid Chromatogr., 16(12), p.2661 - 2672, 1993/00
no abstracts in English
 fuel
fuelNuclear Technology, 90, p.186 - 190, 1990/05
Times Cited Count:7 Percentile:60.93(Nuclear Science & Technology)no abstracts in English
 -HI mixed acid solution
-HI mixed acid solutionUsuda, Shigekazu; Sakurai, Satoshi; Hirata, Masaru; Umezawa, Hirokazu
Sep. Sci. Technol., 25(11-12), p.1225 - 1237, 1990/00
Times Cited Count:1 Percentile:27.36(Chemistry, Multidisciplinary)no abstracts in English
; ; ;
Journal of Nuclear Science and Technology, 24(11), p.931 - 936, 1987/11
Times Cited Count:7 Percentile:60.01(Nuclear Science & Technology)no abstracts in English
; *; ;
Journal of Radioanalytical and Nuclear Chemistry, Letters, 117(6), p.329 - 336, 1987/06
no abstracts in English
; ; *; ;
Journal of Radioanalytical and Nuclear Chemistry, 116(1), p.125 - 132, 1987/01
Times Cited Count:3 Percentile:38.12(Chemistry, Analytical)no abstracts in English
 in HNO
in HNO solution
solutionJournal of Nuclear Materials, 138, p.152 - 154, 1986/00
Times Cited Count:26 Percentile:98.2(Materials Science, Multidisciplinary)no abstracts in English
; ; ; ;
JAERI-M 9530, 17 Pages, 1981/06
no abstracts in English
; ; Shimooke, Takanori; *
JAERI-M 9026, 47 Pages, 1980/09
no abstracts in English
Journal of Radioanalytical Chemistry, 50(1-2), p.133 - 142, 1979/00
no abstracts in English